Atomic Radius Trend: A Simple Guide to How Atoms Change Size
Imagine every single thing in the world is made of tiny, round balls called atoms. Just like you might measure the size of a basketball, scientists measure the size of these atoms. We call this measurement the atomic radius. It is basically the distance from the center of the atom to its outer edge. Knowing the atomic radius trend is like having a secret map of the periodic table. It tells us how these tiny building blocks change as we move around the chart. Some atoms are huge and fluffy, while others are small and tight. When we understand the atomic radius trend, we can predict how chemicals will react with each other in real life. It makes science feel a lot more like solving a giant, invisible puzzle.
Table of Contents
Why Do Atoms Have Different Sizes?
You might wonder why all atoms aren’t just the same size. The answer lies in what is happening inside them. Every atom has a center called a nucleus, which acts like a magnet. This magnet pulls on little particles called electrons that circle around it. The atomic radius trend happens because this “magnet” gets stronger or weaker depending on where the atom sits on the periodic table. If the magnet is very strong, it pulls the electrons in close, making the atom tiny. If there are many layers of electrons, the atom gets much bigger, like adding layers to an onion. Understanding this atomic radius trend helps us see that atoms are not just random circles. They follow very specific rules that help them stay organized.
The Atomic Radius Trend Across a Row
Getty Images
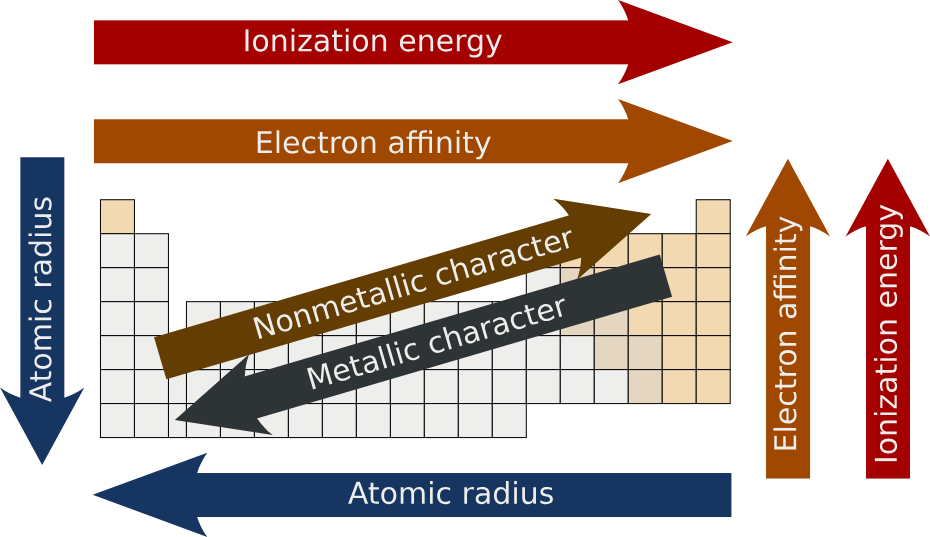
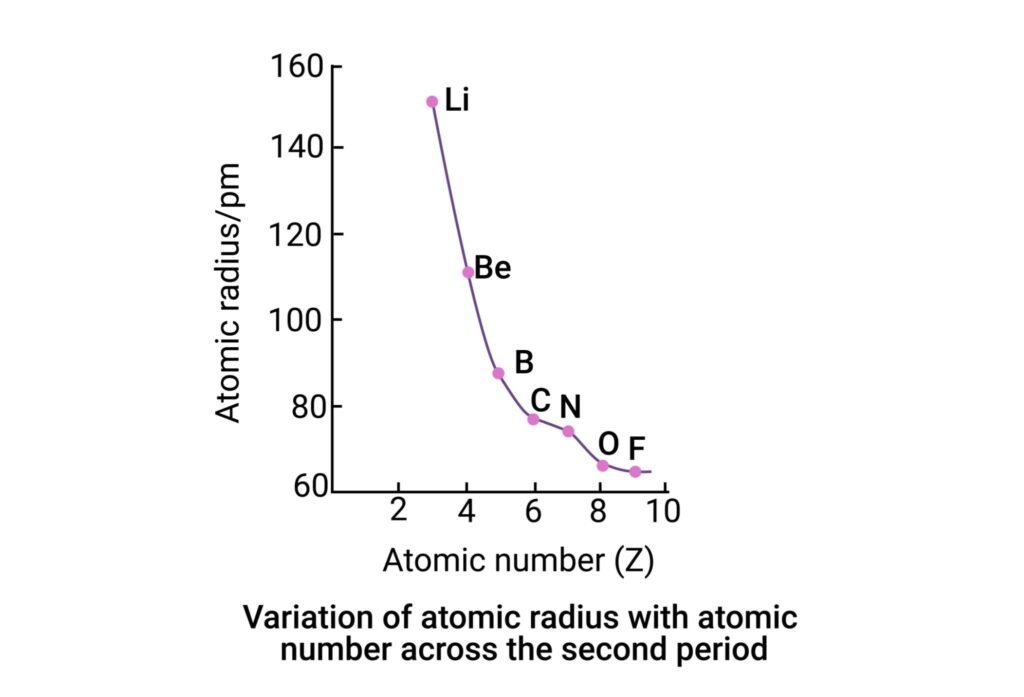
When you look at the periodic table, you see rows that go from left to right. As you move to the right, a strange thing happens to the atomic radius trend. Even though the atoms are getting “heavier,” they actually get smaller! This is because more protons are added to the center magnet. Since the center is stronger, it pulls the outer electrons inward with a lot of force. Think of it like a group of people holding hands in a circle. If the person in the middle pulls everyone closer, the circle gets tighter and smaller. This side-to-side atomic radius trend is one of the most important things to remember for your science class. It proves that more “stuff” inside doesn’t always mean a bigger total size.
Moving Down the Periodic Table
Now, let’s look at what happens when we move from the top of the table to the bottom. In this direction, the atomic radius trend goes the opposite way. The atoms get much larger as you go down! Each step down a column adds a whole new “shell” or layer for the electrons to live in. Imagine putting on five heavy jackets; you would look much bigger than if you were just wearing a t-shirt. This downward atomic radius trend is very consistent. Because the outer electrons are so far away from the center magnet, they aren’t held as tightly. This makes the atom big and a bit “squishy.” Knowing this part of the atomic radius trend explains why elements at the bottom behave differently than those at the top.
How Protons Control the Size
Protons are the tiny parts in the center of an atom that have a positive charge. They are the “bosses” that decide the atomic radius trend. When an atom has more protons, it has more power to pull electrons toward the center. Scientists call this pull “effective nuclear charge.” If the pull is high, the atomic radius trend shows the atom shrinking. If the pull is weak, the atom stays large. It is a constant tug-of-war between the center and the outside. By looking at how many protons are in an element, you can almost always guess the atomic radius trend. It is a very cool way to see how tiny forces create big changes in the physical world around us.
The Role of Electron Shells
Electron shells are like the floors of a tall apartment building. The more shells an atom has, the taller the building is. This is a huge factor in the atomic radius trend. Atoms at the top of the periodic table only have one or two shells, so they are quite small. But atoms at the bottom can have seven shells! This extra space makes the atomic radius trend jump up significantly. Even if the center magnet is strong, it can’t overcome the fact that there are so many “floors” of electrons stacked on top of each other. When you think about shells, the atomic radius trend becomes very easy to visualize. Just remember: more layers always equal a bigger atom.
What is Electron Shielding?
Shielding sounds like something a knight would do, and in a way, it is! In an atom, the inner electrons block the center magnet’s pull from reaching the outer electrons. This “shielding effect” plays a big role in the atomic radius trend. When there are many inner electrons, the outer ones don’t feel the pull very much, so they wander further away. This makes the atom expand. Without shielding, the atomic radius trend would look very different. It is like trying to hear a teacher from the back of a noisy room; the sound (or the pull) just doesn’t reach you as well. This is why atoms with many layers stay so large and fluffy.
Quick Reference Comparison Table
| Element Name | Position on Table | Atomic Size | Why? |
| Hydrogen | Top Left | Very Small | Only one electron shell. |
| Lithium | Left Side | Medium | Starts a new shell. |
| Neon | Right Side | Small | Strong center pull shrinks it. |
| Cesium | Bottom Left | Very Large | Many shells and weak pull. |
| Fluorine | Top Right | Tiny | Strong pull and few shells. |
Real-Life Examples of Atom Sizes
You might think the atomic radius trend is just for textbooks, but it affects real things! For example, Cesium atoms are very large. Because they are big, their outer electrons can jump away easily, which makes Cesium very reactive in water. On the other hand, Fluorine is tiny and holds its electrons very tightly. This difference in the atomic radius trend is why some chemicals are dangerous and others are stable. Even the salt you put on your food depends on the sizes of Sodium and Chlorine atoms fitting together. By studying the atomic radius trend, scientists can create new medicines and stronger materials for building cars or phones.
Comparing Cations and Anions
Sometimes atoms lose or gain electrons, and they become “ions.” This changes the atomic radius trend instantly. If an atom loses an electron (a cation), it usually gets smaller because the remaining electrons are pulled in tighter. If an atom gains an electron (an anion), it gets bigger because the new electron pushes the others away. This “ion version” of the atomic radius trend is a fun twist on the basic rule. It’s like a balloon; if you let air out, it shrinks, and if you blow more in, it grows. Seeing how ions change helps us understand how electricity moves through batteries and even through your own body!
How to Predict Atom Sizes Easily
If you are studying for a test, here is a simple trick for the atomic radius trend. Just remember “Down and Left.” If you move down the table, the atoms get bigger. If you move to the left of the table, the atoms get bigger. Therefore, the biggest atoms are always in the bottom-left corner, and the smallest are in the top-right corner. This simple rule makes the atomic radius trend easy for anyone to master. You don’t need to memorize every single number. Just look at the shape of the table and follow the arrows. This trick has helped many students get an A+ in their chemistry lessons.
Frequently Asked Questions
1. Does atomic radius mean the weight of the atom?
No! The atomic radius trend is about size (volume), not weight (mass). An atom can be heavy but still very small if its electrons are pulled in tight.
2. Which element has the largest atomic radius?
Francium is usually considered the winner of the atomic radius trend because it is at the very bottom-left of the periodic table.
3. Why does Helium have such a small radius?
Helium is at the top-right. It has a strong pull for its size and very few electrons, making it one of the smallest atoms in the atomic radius trend.
4. How do scientists measure such tiny things?
They use special X-rays and math to see how far apart the centers of two atoms are when they are touching.
5. Does the atomic radius trend change in space?
The rules of chemistry stay the same everywhere in the universe, so the atomic radius trend works on Mars just like it does on Earth!
6. Why is this important for me to learn?
Understanding the atomic radius trend helps you understand how the world is built at its most basic level, which is the start of all great inventions.
Conclusion: Mastering the Map of Atoms
Learning about the atomic radius trend is the first step to becoming a science pro. It shows us that the universe follows a beautiful, organized pattern. From the tiny Fluorine atom to the giant Cesium atom, every element has its place. By remembering that atoms get bigger as you go down and smaller as you move right, you can predict how almost any element will behave. Chemistry isn’t just about mixing liquids in a lab; it’s about understanding these tiny shapes and sizes. So next time you see a periodic table, remember the atomic radius trend and how it explains the hidden world of the very small!